As if the people in Scotland are not ugly and retarded enough, now they want to breed even uglier retards!
Namely the evil limbless Thalidomides !!!!


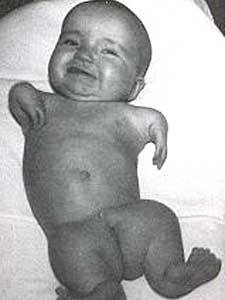

THIS artcile has me in shock!!!
Namely the evil limbless Thalidomides !!!!




THIS artcile has me in shock!!!

Green light for thalidomide drug
Doctors have been given the green light to prescribe the drug thalidomide in Scotland.
The go-ahead was given by the Scottish Medicines Consortium (SMC) which scrutinises drugs on cost-effectiveness grounds.
Thalidomide was linked to thousands of cases of birth defects worldwide 50 years ago.
About 12,000 people whose mothers took thalidomide to prevent morning sickness were born with severe deformities in Europe, Japan and Canada. The connection with the drug was made in 1961 and it was withdrawn from sale in the UK that year.
Last year, however, the European Medicines Agency recommended thalidomide for the treatment of a rare bone marrow cancer.
The effect of the SMC decision is to enable doctors in Scotland to prescribe the drug without first having to argue a case for it on clinical grounds to their health boards.
Up to now, it could only be used for "off-label" prescribing, after doctors had convinced their health board that it was suitable in exceptional cases.
The SMC said the drug was now accepted for use, in combination with other drugs, as a first-line treatment for certain patients with untreated multiple myeloma, a rare cancer of a type of white blood cell.
Doctors have been given the green light to prescribe the drug thalidomide in Scotland.
The go-ahead was given by the Scottish Medicines Consortium (SMC) which scrutinises drugs on cost-effectiveness grounds.
Thalidomide was linked to thousands of cases of birth defects worldwide 50 years ago.
About 12,000 people whose mothers took thalidomide to prevent morning sickness were born with severe deformities in Europe, Japan and Canada. The connection with the drug was made in 1961 and it was withdrawn from sale in the UK that year.
Last year, however, the European Medicines Agency recommended thalidomide for the treatment of a rare bone marrow cancer.
The effect of the SMC decision is to enable doctors in Scotland to prescribe the drug without first having to argue a case for it on clinical grounds to their health boards.
Up to now, it could only be used for "off-label" prescribing, after doctors had convinced their health board that it was suitable in exceptional cases.
The SMC said the drug was now accepted for use, in combination with other drugs, as a first-line treatment for certain patients with untreated multiple myeloma, a rare cancer of a type of white blood cell.
Comment